 (Oxygen Gas, also called Elemental Oxygen) (Dinitrogen Tetrahydride or Hydrazine or Diamine) (Nitrogen Gas, also called Elemental Nitrogen) (Hydrogen Carbonate Ion or Bicarbonate Ion) (Hydrogen Peroxide or Dihydrogen Dioxide) The octet rule is satisfied for each of the 5 chlorine atoms, but it is exceeded for the phosphorus atom.Click the Chemical Formula to see the Lewis Structure Acetone This molecule has 5 chlorine atoms covalently single-bonded to the central atom, phosphorus. Phosphorus pentachloride (PCl 5) breaks the octet rule by having 5 bonding pairs around the central atom. 
This allows the octet rule to be satisfied for all 3 atoms. For example, carbon dioxide (CO 2) has 2 oxygens covalently double-bonded to the central atom, carbon.Some large atoms such as phosphorus can break the octet rule.This can help you determine the number of bonds that will be between the central atom and the other atoms because each bond represents 2 electrons. When the central atoms bonds to the other atoms, the lowest energy configuration is one that will satisfy the octet rule (in most cases). As a general (but not all-exclusive) rule, atoms like to be surrounded by 8 valence electrons (the octet rule). Therefore, 2 of the electrons are required to make a double bond between the atoms so the octet rule is met for both.Ĭonsider the valence electrons of the central atom. This signifies that more than 1 bond is required between the 2 oxygens. For O2, one oxygen has 8 electrons (so the octet rule has been met), but the other has only 6 (so the octet rule has not been met). To determine if the octet rule has been met, use dots to represent the valence electrons around each atom.For instance, O2 (oxygen gas) has 6 valence electrons. 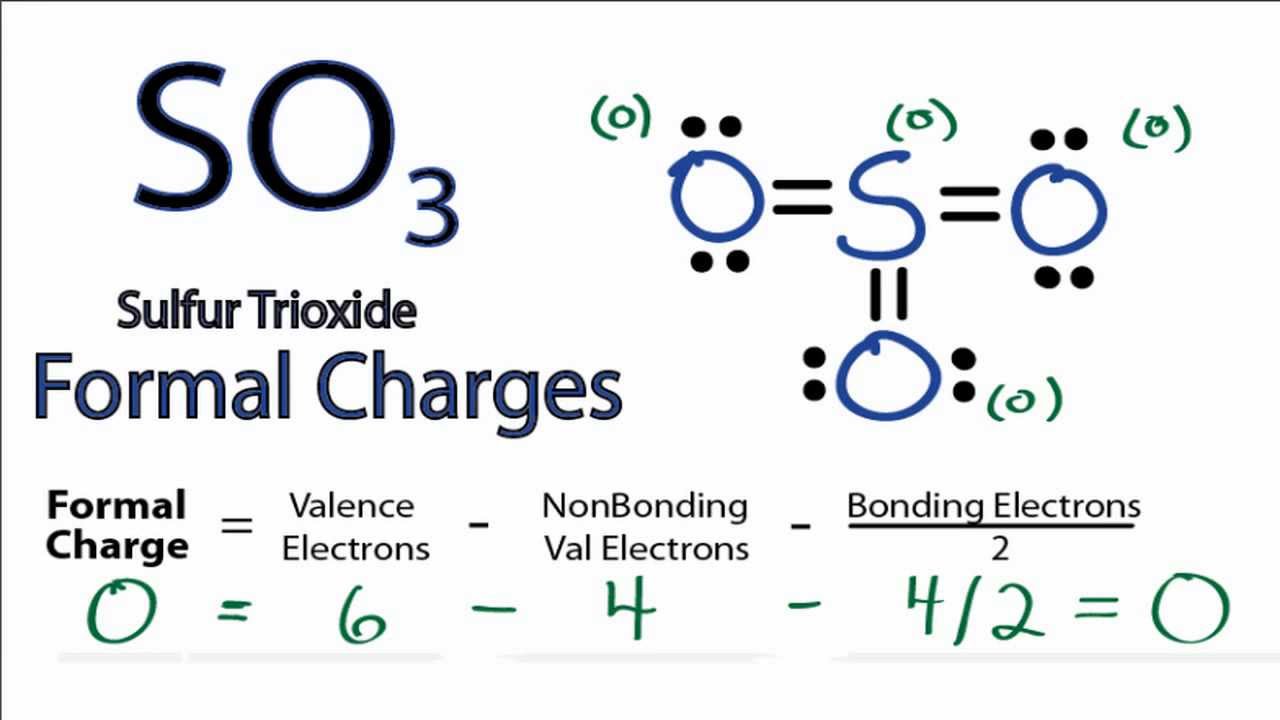
To determine how many electrons each atom will have, find out how many valence electrons are in the molecule, multiply that by 2 (each bond involves 2 electrons), and then add the number of unshared electrons. Generally, this will be dictated by the octet rule, or each atom’s desire to reach a full valence shell with 8 electrons (or in the case of hydrogen, 2 electrons). 
Atoms can be held together by a single, double, or triple bond. 
Determine the degree of the bond between the 2 atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
